Risk and hazard assessment of chemicals
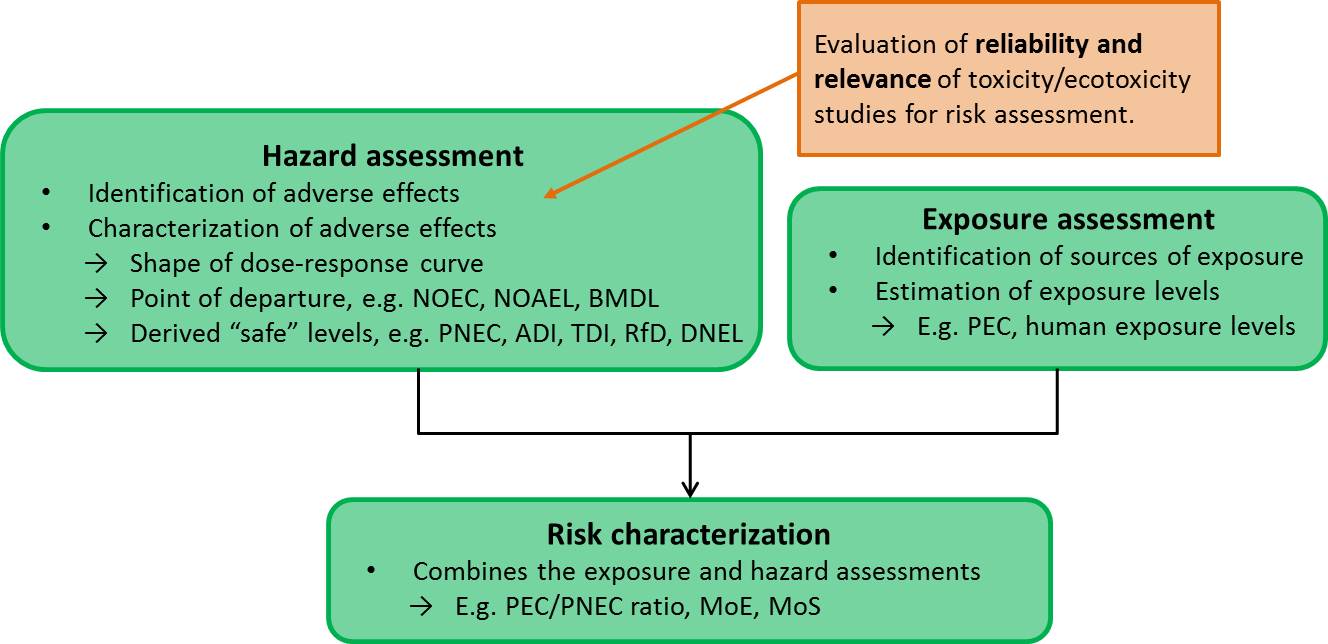
Risk and hazard assessment of chemicals is a process performed with the overall objective to protect human health and the environment. It entails identifying and characterizing the risk of adverse health effects (health risk assessment) or effects in the environment (environmental risk assessment) occurring at current exposure levels. Risk assessment consists of three main parts; hazard assessment, exposure assessment and risk characterization (see figure below).
The hazard assessment aims to identify and characterize the adverse effects of the compound and establish a point of departure, such as a no observed effect concentration (NOEC) for environmental risk assessment, or a no observed adverse effect level (NOAEL) or a lower confidence limit of a benchmark dose (BMDL) for health risk assessment.
From these points of departure “safe” levels can be derived. In environmental risk assessment a predicted no effect concentration (PNEC) is commonly established. In health risk assessment an acceptable or tolerable daily intake (A/TDI), a reference dose (RfD) or derived no effect level (DNEL) may be determined in this step.
In this process available toxicity and/or ecotoxicity data (QSAR, in vitro, in vivo and epidemiological data) for the substance are reviewed and evaluated as to their reliability and relevance for inclusion as key or supporting evidence in risk assessment.
In the exposure assessment possible sources of exposure are identified and exposure levels in the environment, e.g. predicted environmental concentration (PEC), or in human populations are measured or calculated.
In the risk characterization step the hazard and exposure assessments are combined to draw conclusions about risk. In environmental risk assessment a PEC/PNEC ratio is commonly calculated. For health risk assessment a comparison is made to see if estimated human exposure levels exceed the derived “safe” levels, or the point of departure may be divided by the estimated exposure to calculate margins of safety (MoS), sometimes also referred to as margins of exposure (MoE).
The structure of risk assessment: